The cross-functional imaging innovation area comprises several imaging technologies that are used in research and diagnostics in the preclinical and clinical domains. These include classical histology and immunohistochemistry as well as state-of-the-art technologies such as non-invasive in vivo fluorescence and luminescence imaging in animal models, radiation-free examination of microcirculation in the hands (Xiralite platform) and High Content imaging for in vitro assays.
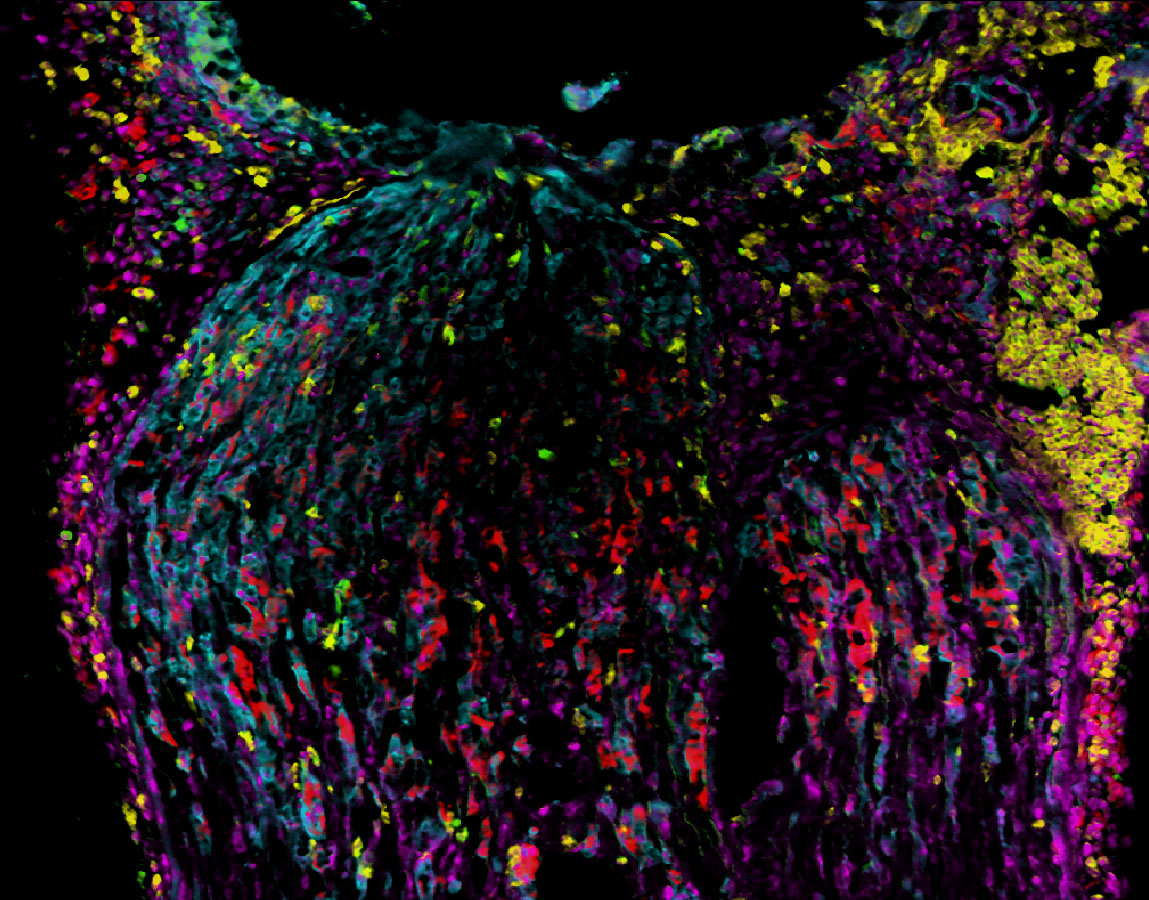
A core technology used is the »SMILE« (Sequential Multiple Immunohistochemistry for Localization- and Expression analysis) system for the multiple sequential immunohistochemistry and visualization of up to 100 proteins in the same sample. The images are subsequently analyzed to identify and quantify cellular microenvironments in a tissue as well as the effects of compounds on immune responses. This rapid and cost-effective multiplexing technique also allows for significant reduction in the amount of tissue isolated from biopsies and individualized therapy recommendations.
Core Competencies:
- SMILE System for multiplexing immunohistochemistry
- In vivo fluorescence- and luminescence-imaging (IVIS Spectrum)
- In vitro High-Content imaging using confocal fluorescence microscopy (e.g. ImageXpress)
- Single cell calcium imaging
- Radiation-free investigation of microcirculation of the hands (Xiralite)
 Fraunhofer Institute for Translational Medicine and Pharmacology ITMP
Fraunhofer Institute for Translational Medicine and Pharmacology ITMP