Start of new EU research project HIPPOCRATES: promoting early identification and improving outcomes for patients with psoriatic arthritis
Funded by the Innovative Medicines Initiative (IMI), the Fraunhofer Cluster of Excellence Immune-Mediated Diseases CIMD, together with 25 European partners from research, pharmaceutical companies, SME’s and patient organizations, is researching a poorly understood disease that affects millions of people. By looking into the disease mechanisms of psoriatic arthritis, the 26 European partners collaborating in the new research project HIPPOCRATES aim at improving diagnostic and therapeutic options for patients living with this condition. Through gaining a better understanding of the complex interplay between clinical and environmental factors, genotype and molecular pathways, the team aims to enable earlier diagnosis and a more accurate prediction of disease progression. This will revolutionize treatment and deliver profound patient benefits.
FRANKFURT. Psoriatic arthritis (PsA) is a chronic immune-mediated inflammatory disease that affects joints and other components of the musculoskeletal system, together with skin involvement, in an estimated 5-10 million individuals in the EU. The symptoms of the disease, including pain, joint stiffness and fatigue, can impact on many aspects of life including function and productivity. Overall, it is increasingly recognized that PsA is associated with multiple comorbidities, particularly those affecting mental health such as depression and those which promote the development of accelerated atherosclerosis and contribute to the observed increase in cardiovascular morbidity and mortality. PsA most commonly develops on a background of established skin and/or nail psoriasis, however it can be difficult to diagnose as there are no diagnostic criteria or laboratory tests available. This can contribute to diagnostic delay and poor outcomes. PsA is characterized by considerable heterogeneity with regards to clinical features, disease progression and response to targeted therapies. Future treatments will need to focus on earlier disease stages and be selected on the basis of detailed patient molecular profiling so as to limit poor long-term outcomes and possibly prevent the development of PsA altogether.
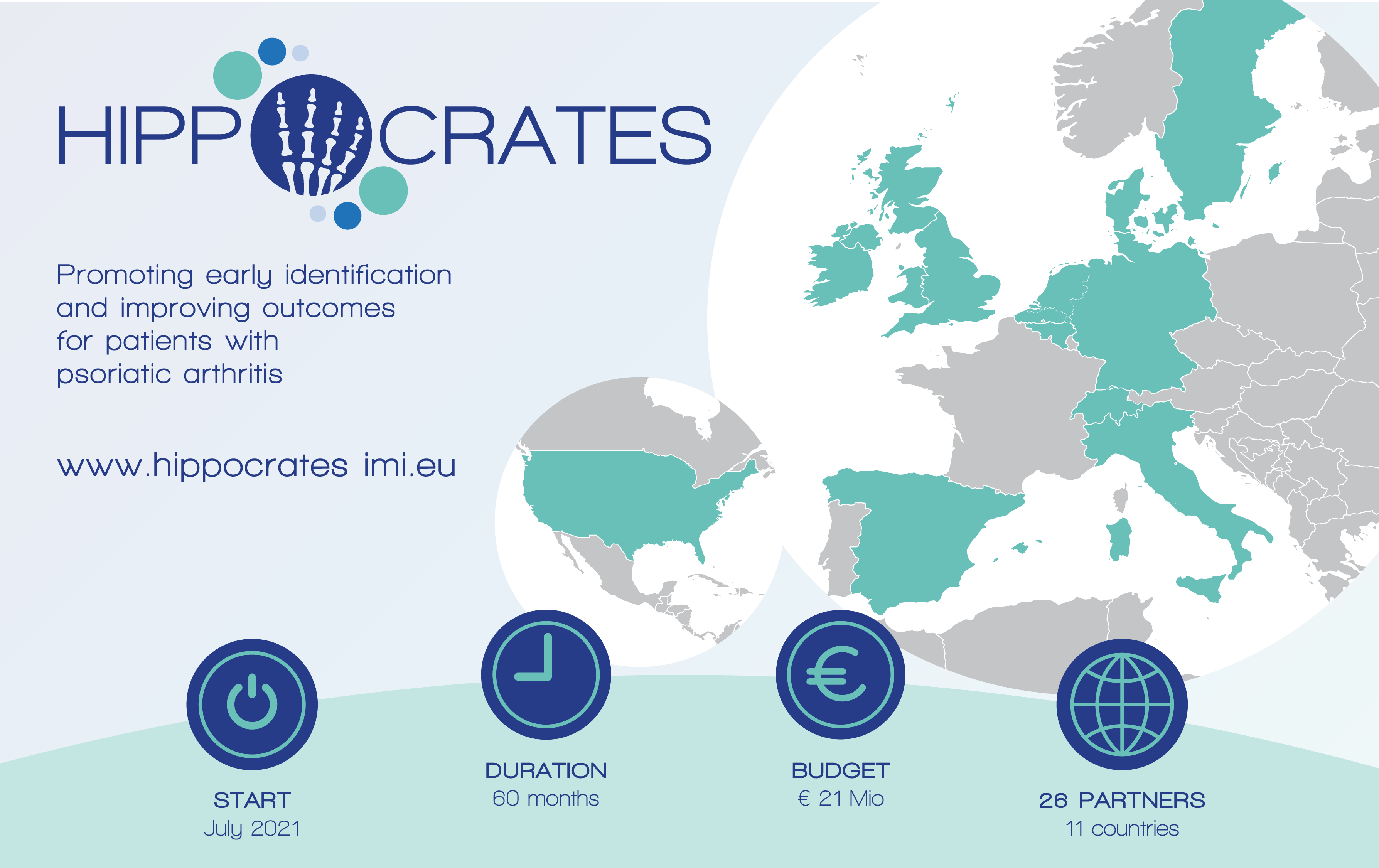
Three Fraunhofer institutes of the Fraunhofer Cluster of Excellence Immune-Mediated Diseases CIMD (Fraunhofer ITMP, IAIS and IGD) participate in the European HIPPOCRATES (Health Initiatives in Psoriasis and PsOriatic arthritis ConsoRTium European States) consortium, which was launched, in order to identify patients at risk of developing PsA, and to develop diagnostic methods and personalized approaches to treat patients. Formed as a transdisciplinary consortium, the project team comprises 26 partner institutions from Belgium, Denmark, Germany, Ireland, Italy, Spain, Sweden, Switzerland, the Netherlands, the United Kingdom and the United States of America. Led by University College Dublin, the partners contribute a diverse range of backgrounds including clinical, scientific, data analytics, ethics, patient participation as well as SME and pharmaceutical industry expertise in pursuit of the ambitious goals set for the HIPPPOCRATES project. The project will run for a period of five years with a total budget of EUR 21 million provided by the Innovative Medicines Initiative (IMI 2), a Joint Undertaking of the European Union and the European Federation of Pharmaceutical Industries and Associations (EFPIA). Of the total budget, 50% are contributed by the EFPIA partners (Novartis [EFPIA lead], UCB [EFPIA Co-lead], Pfizer and BMS) and 50% by the EU.
“We anticipate that the advances provided by HIPPOCRATES will result in significant new developments that improve patients’ quality of life” says Prof. Oliver FitzGerald, Newman Clinical Research Professor at University College Dublin, Conway Institute for Biomedical and Biomolecular Research, Ireland coordinator of the HIPPOCRATES consortium. Co-coordinator, Prof. Stephen Pennington, Professor of Proteomics at University College Dublin and also in the Conway Institute notes “The advances will include the identification of sub-populations and endotypes, the validation of existing and identification of new biomarkers, improved imaging options and the development of a sustainable infrastructure for future PsA research.” “HIPPOCRATES offers the great promise of powerful new tools to advance both early diagnosis and treatment of patients with PsA. Additionally, and to further its effectiveness, HIPPOCRATES retains the focus on the patient, involving Patient Research Partner’s within all aspects of the project” adds Denis O’Sullivan (Patient Representative Arm of GRAPPA-EU). “This public-private partnership is a great opportunity to decipher this highly heterogeneous disease, and to enable the development of novel PsA therapies and treatment strategies including precision medicine approaches” adds Dr. Christine Huppertz, Senior Principal Scientist in the Disease Area Autoimmunity, Transplantation and Inflammation at Novartis, and EFPIA lead of the consortium.
In order to achieve its goals, the HIPPOCRATES project will set up a single integrated database combining the cohorts and datasets of the most important European PsA studies and establish a Europe-wide library of relevant clinical biosamples. HIPPOCRATES will also establish a large, prospective, observational study of 25,000 patients with psoriasis who will be recruited and followed on-line for development of PsA, with patient-centric blood sampling at defined intervals. Furthermore, the team of experts will evaluate and validate newly discovered biomarker signatures for the early diagnosis of PsA, for the identification of psoriasis patients at risk of developing PsA, for the identification of PsA patients at highest risk of damage progression and for personalized or stratified treatment strategies so as to maximize treatment response. Overall, HIPPOCRATES places particular emphasis on the involvement of patients, clinicians, primary care practitioners, regulators, SMEs (ATTUROS Limited, Oxford Biodynamics Limited and NEOTERYX Limited) and relevant large industry to meet the needs of all stakeholders and to maximize the project’s impact.
Fraunhofer ITMP, in cooperation with the Department of Rheumatology of the University Hospital Frankfurt, was involved at an early stage in planning the content and institutional composition of the consortium. Together with its Fraunhofer CIMD partner institutes Fraunhofer IAIS and IGD, it will lead two of the seven HIPPOCRATES work packages, covering “Early Diagnosis of PsA” and “Data Integration and Analysis”. For the early diagnosis of PsA, the Fraunhofer team will determine clinical, imaging, and molecular disease features and capture them in a combined context to derive algorithms for PsA diagnosis. Data integration and analysis will involve the use of machine learning to model an AI-based risk score of progression from psoriasis to PsA, as well as a risk score for rapid bone-damaging disease progression and a response score to predict response to a given treatment.
“HIPPOCRATES offers us the opportunity to collaborate with leading European experts to gain a deeper understanding of PsA in order to improve the mobility and quality of life of many affected patients,” says PD Dr. Frank Behrens, Head of Clinical Research at Fraunhofer ITMP and member of the Fraunhofer CIMD Board of Directors. “I am confident that in our project we will discover new markers that will enable early detection of PsA, improve treatment response and possibly even open up avenues for disease prevention. With the unique combination of the diverse expertise represented in Fraunhofer Health Research, from medicine to multi-omics, and image processing to data sciences, we can make an important contribution to manage the debilitating PsA disease.”
Project Key Facts
Full name:
HIPPOCRATES – Promoting Early Identification and Improving Outcomes in Psoriatic Arthritis
Start date:
1st July 2021
Duration:
60 months
Budget:
21 Mio. €
Coordinator:
University College Dublin, Conway Institute for Biomedical and Biomolecular Research, Irland
Webseite:
Project Partners
Belgium
- Katholieke Universiteit Leuven
- The European Institute for Innovation through Health Data
- UCB Biopharma SRL
- VIB VZW
Denmark
- Rigshospitalet
Germany
- Eurice - European Research and Project Office Gmbh
- Fraunhofer-Gesellschaft zur Förderung der Angewandten Forschung e.V.
- Friedrich Alexander University Erlangen-Nürnberg
Ireland
- ATTUROS Limited
- University College Dublin (UCD)
Italy
- Università Cattolica Del Sacro Cuore
Netherlands
- Academisch Medisch Centrum Bij De Universiteit Van Amsterdam
- Stichting Group for Research and Assessment of Psoriasis and Psoriatic Arthritis EU
Spain
- Consorci Institut D'investigacions Biomediques August Pi I Sunyer
Sweden
- Kungliga Tekniska Hoegskolan
Switzerland
- European League Against Rheumatism
- Novartis Pharma AG
- SIB Institut Suisse de Bioinformatique
United Kingdom
- King's College London
- Neoteryx Limited
- Oxford Biodynamics Limited
- Pfizer Limited
- University of Glasgow
- University of Manchester
- University of Oxford
United Staates of America
- Bristol-Myers Squibb Company Corp
Patient Organisations involved in the project:
- EULAR PARE (The European Alliance of Associations for Rheumatology)
- GRAPPA-EU (Group for Research and Assessment of Psoriasis and Psoriatic Arthritis)
- EUROPSO (European Umbrella Organisation for Psoriasis Movements)
Contact
Project Coordinators:
University College Dublin, Conway Institute for
Biomedical and Biomolecular Research, Ireland
Prof. Oliver FitzGerald
Newman Clinical Research Professor
Phone: +35 3872486687
University College Dublin, Conway Institute for
Biomedical and Biomolecular Research, Ireland
Prof. Stephen Pennington
UCD Professor of Proteomics
Phone: +35 1 716 6783
Project Management:
EURICE GmbH
Martha Whiteman
Phone: +49 30 37 44 15 - 832
EFPIA Lead:
Senior Principal Scientist, Novartis Pharma AG
Dr. Christine Huppertz
Phone: +41 79 292 6128
EFPIA Co-Lead:
Head of Medical Affairs Immun-Dermatology, UCB Pharma
Dr. Owen Davies
Phone: +44 7768 275135
 Fraunhofer Institute for Translational Medicine and Pharmacology ITMP
Fraunhofer Institute for Translational Medicine and Pharmacology ITMP