Alternative SARS-CoV-2 infection route via cerebral vessels identified
Neurological complications associated with COVID-19 have been observed frequently. Although SARS-CoV-2 may be abundant in brain tissue of patients, its pathways of entry and the resulting consequences are not well understood. A team of researchers from the Fraunhofer Institute for Translational Medicine and Pharmacology ITMP and the University Medical Center Hamburg-Eppendorf (UKE) has now shown that cells of the blood-brain barrier (BBB) can be infected in vitro by the SARS-CoV-2 pathogen. The BBB is an important interface between the peripheral vasculature and the central nervous system. In addition, a test system based on human pluripotent stem cells has been developed to screen therapeutics for their effectiveness in attenuating this infection. The study results were published today in the journal Stem Cell Reports.
Hamburg. Fraunhofer scientists have already been developing preclinical models and, assisted by them, drug candidates for the treatment of COVID-19 since February 2020. “The ‘Fraunhofer vs. Corona’ action program has enabled us to focus our resources and
expertise on the pandemic and bring them into application” says Dr. Ole Pless, a senior scientist at Fraunhofer ITMP and also the laboratory head of a visiting group at the UKE's Center for Molecular Neurobiology Hamburg (ZMNH). “In addition, we developed the relevant cell models in a project funded by the German Federal Ministry of Education and Research, which we were able to bring directly to application during the pandemic.”
“Based on our extensive knowledge of COVID-19 pathology, including central nervous system pathology, we were able to describe precise molecular changes at the blood-brain barrier. These were confirmed in cell models provided by Fraunhofer”, said the study's first author, Priv.-Doz. Dr. Susanne Krasemann from the Institute of Neuropathology at the UKE. The study was conducted with the participation of several UKE institutes and other international partners.
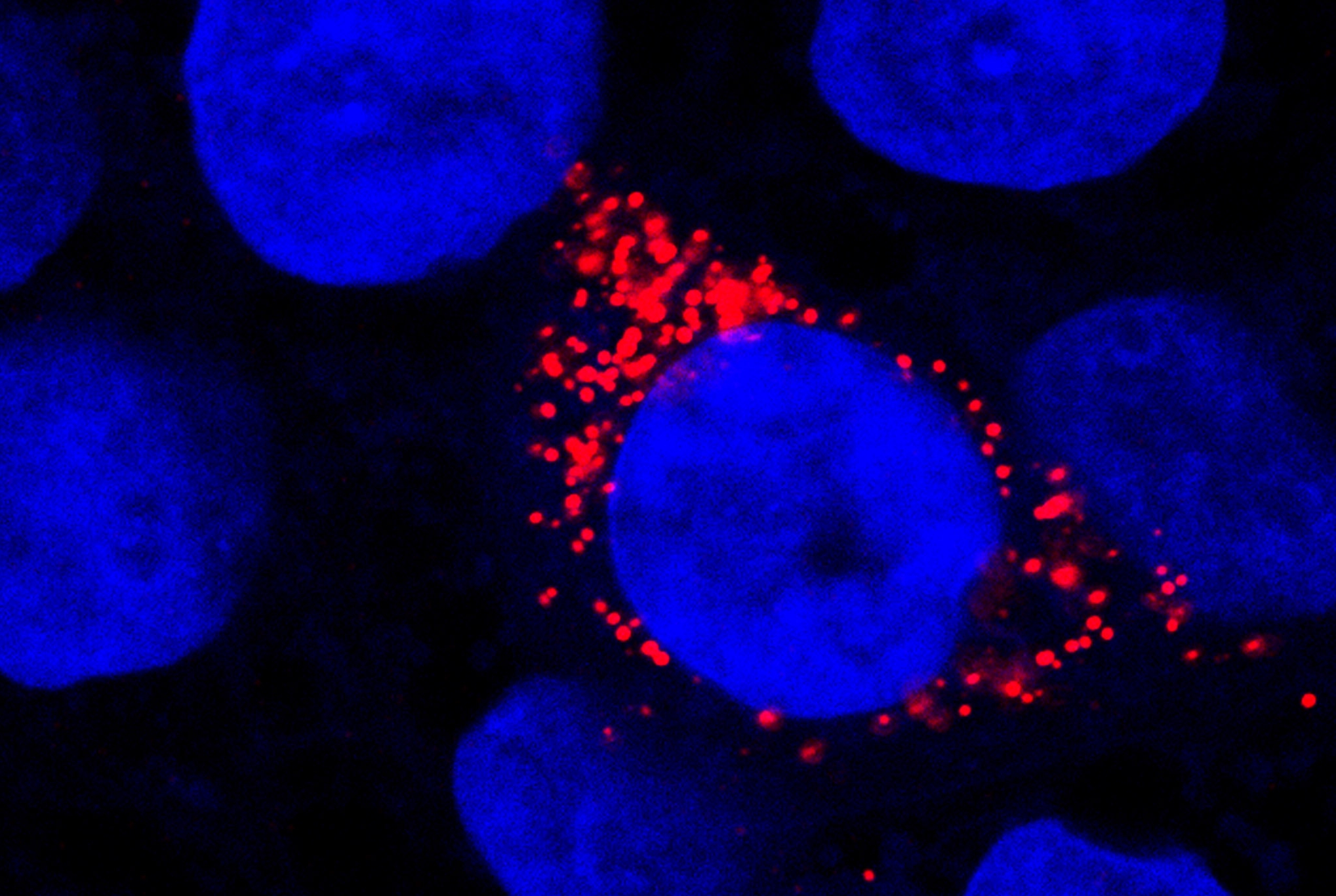
COVID-19: Characteristics of brain pathology can be reproduced in cell culture
Extensive analysis of post-mortem brain tissue from COVID-19 patients demonstrated upregulation of interferon-gamma signaling pathways, a hallmark of viral defense, in the neurovascular unit of the BBB. By using pluripotent stem cells, the study authors were able to mimic this susceptibility of brain capillary endothelial cells to SARS-CoV-2 infection in the laboratory. In an in vitro test system, they were able to observe the replication of the virus in the cells and its transport across the BBB. This model was subsequently applied to test compounds that prevent virus entry across the BBB, such as specific protease inhibitors. Thus, a platform was created for testing drugs that could prevent SARS-CoV-2 pathogens from entering the central nervous system and mitigate the resulting neurological consequences.
A key challenge for society
“A large number of scientists from various UKE departments have been conducting research on the SARS-CoV-2 pathogen since the beginning of the pandemic. We are coordinating the various dimensions of research relevant to the pandemic and see great importance in collaborations with other research institutions such as the Fraunhofer ITMP. The interaction between clinical and basic research is particularly close to our hearts”, says Prof. Dr. Blanche Schwappach-Pignataro, Dean of the Faculty of Medicine and member of the UKE Board of Directors.
“Our aim at the recently founded Fraunhofer Institute ITMP is to translate the most promising ideas from basic biomedical research into applications for the benefit of
patients and society. It is precisely the close connection between applied research and everyday clinical practice, as practiced at our locations in Frankfurt, Göttingen, Penzberg/Munich, Berlin and with the UKE in Hamburg, that is key to developing new therapies”, says Prof. Dr. Dr. Gerd Geißlinger, director of Fraunhofer ITMP.
Publication
Krasemann et al., The blood-brain barrier is dysregulated in COVID-19 and serves as a CNS entry route for SARS-CoV-2. Stem Cell Reports, 2022.
DOI: https://doi.org/10.1016/j.stemcr.2021.12.011
 Fraunhofer Institute for Translational Medicine and Pharmacology ITMP
Fraunhofer Institute for Translational Medicine and Pharmacology ITMP